
With the current high level of interest in patches to deliver drugs across the skin, the concept of external applications for delivery of medicinal substances is becoming widely accepted. This study evaluated the potential for transdermal absorption of castor oil, a treatment modality recommended by Edgar Cayce. Specific epoxydicarboxylic acids are known to be excreted via the urine when castor oil is administered orally. Comparison of the amounts of these substances in the urine of subjects given oral and transdermal administrations of castor oil may provide evidence of transdermal absorption of castor oil and provide clues to its metabolism in the system. Three volunteers were given castor oil via abdominal hot pack and oral administration. Urine samples were collected before and after the interventions. Oral administration of castor oil resulted in high levels of excretion of the epoxydicarboxylic acids in all the subjects, results similar to a previous study. In contrast, the level of urinary epoxydicarboxylic acids in the external application (abdominal pack) sessions did not vary greatly from the relatively low endogenous levels of these molecules that are normally present. If the castor oil was absorbed through the skin and metabolized in the system, it did not have the effect of significantly increasing the excretion of the specific metabolic byproducts associated with ingestion of castor oil. The implications of these findings are discussed and directions for future research are considered. Introduction Castor oil has a long history as a medicinal agent in folk healing and mainstream medicine. Its primary function has been as an oral laxative. In his book The Oil That Heals (1993), McGarey detailed numerous case reports supporting the therapeutic efficacy of externally applied castor oil packs for a wide range of disorders. McGary's review relies heavily on the Edgar Cayce legacy. The work of Edgar Cayce, an important figure in the development of holistic medicine in the modern era (Callan, 1979; Mein, 1989), places great significance on healing properties of castor oil applied externally as a pack. The typical application recommended by Cayce is that of a hot abdominal pack placed over the right side of the abdomen covering the liver, upper small intestines, cecum, and ascending colon. One of the major drawbacks for using the Cayce regimen for castor oil
packs is that they tend to be messy and difficult to apply. With
the current high level of interest in patches to deliver drugs across the
skin, the concept of external applications for delivery of medicinal substances
is becoming widely accepted. The intent of this study was to develop
a reliable means for measuring levels of transdermal absorption of externally
applied castor oil. Identifying one or more chemical markers could
then allow for comparison of Cayce's typical application with more modern
systems of dermal delivery that tend to be less messy and more practical.
A previous study done by Hagenfeldt et al. (1986) provided a conceptual framework for our research design. In the Hagenfeldt et al. study, castor oil was administered orally to three healthy volunteers. Urine was collected for several hours and analyzed using capillary gas chromatography. The analysis revealed elevated levels of the products of breakdown of castor oil by the liver (specific epoxydicarboxylic acids that are normally found in urine in very small amounts). Thus, the presence of high levels of these acids is evidence for absorption and metabolism of orally administered castor oil. Using the data and conceptual model from the Hagenfeldt et al. study, we designed a study that included both oral and external (abdominal pack) administration of castor oil. By comparing the levels of the epoxydicarboxylic acids in the urine following external and oral applications, we hoped to be able to reliably quantify the level of absorption of the oil through the skin. Methods Design This preliminary study utilized a within-subjects design with pre and post intervention data collection. Subjects
Procedures
The procedure for the abdominal castor oil pack sessions is documented in Appendix A. Since the level of heat used in the abdominal castor oil pack may be a factor in transdermal absorption, three levels of heat were applied using electrical heating pads (50 watts, 100 watts, and 200 watts). The following sequence of heating was used in the first stage of the project:
First Series Second Series
All urine samples were tested for the presence and concentration of two epoxydicarboxylic acids (epoxyoctanedioic acid and epoxydodecanedioic acid) and ricineolic acid. The urine samples were analyzed by Alturas Analytics, Inc. (Moscow, ID) using the capillary gas chromatography as described by Hagenfield et al (1986). The sequence of interventions and sampling for Stage 1 is provided in Table 1 (Appendix B). The second stage of the project was essentially a focused and scaled down version of the first stage to confirm the findings and decrease sources of error. A different laboratory (EnviroTest Laboratories, Edmonton, AB, Canada) was used to minimize laboratory practices as a source of variability. The highest level of heating pad was used for all sessions to facilitate maximum penetration of the castor oil into the skin. Dietary oils of a similar composition to castor oil were eliminated (e.g., olive oil) at least one week prior to and during the second stage. Since no detectable levels of ricineolic acid was found in any of the samples in stage 1, this substance was not included in the lab assays for stage 2. The sequence of interventions and sampling for Stage 2 is provided in Table 2 (Appendix B). Results For the oral administration of castor oil in either of the two amounts, there was a high level of excretion of the epoxydicarboxylic acids in all the subjects, results similar to the Hagenfeldt et al. study. In contrast, the level of urinary epoxydicarboxylic acids in the external application (abdominal pack) sessions did not vary greatly from the relatively low levels of these molecules normally present. The amount of heat used did not affect the outcome. Discussion Considering how permeable the skin is to a wide range of substances, it would be truly amazing if no significant amounts of castor oil were absorbed after an hour and a half of contact with high heat. Yet we were unable to show this based on measuring the products produced in the liver and excreted in the urine, which are very evident when castor oil is taken orally. We took care to increase the chances of transdermal absorption in Stage 2 by increasing the level of heat to the maximum (200 watt heating pads), prolonging all castor oil sessions to one and one-half hours in duration, and avoiding possible confounding dietary sources of oils similar to castor oil (especial olive oil). Apparently these adjustments did not have a notable affect on the outcome that is essentially in agreement with the Stage 1 results. The Cayce readings do consistently maintain that small amounts of substances frequently have greater therapeutic effects than large doses. This concept is similar to the homeopathic view on the relative potency of dosages wherein small amounts of chemicals can have a greater or different effect than large amounts. Perhaps a small amount of castor oil absorbed through the skin has a different effect than the same amount absorbed through the gut. When taken orally, castor oil is broken down with powerful enzymes in the small intestine before being sent to the liver for further processing. The raw castor oil could have a local effect directly on the underlying tissues before cycling through the liver for processing, which may take longer without the benefit of intestinal enzymes. Thus, low levels of castor oil could have a therapeutic effect without producing a spike in urinary epoxydicarboxylic acids. To help clarify our findings and give us clues as to how to proceed with the next phase of investigation, we contacted the International Castor Oil Association and several of its members requesting data or insights into the possible transdermal properties of castor oil. So far we have not received any useful suggestions for the further exploration of how castor oil is absorbed through the skin and metabolized in the system. One possibility for further inquiry is to see if ricinoleic acid, the major component of castor oil, is found in the blood immediately after the application of a castor oil pack. If it is not, then apparently the castor oil does not cross the skin barrier. If it is found in the blood, but the breakdown products are not found in the urine in detectable amounts (as we have already determined), then it is metabolized in a way not yet understood. This could offer significant opportunities for further research on the biochemistry and metabolism of castor oil and its relation to healing. Another possibility noted by McGarey is that castor oil is assimilated into the system vibratorially. Pondering the idea that everything is composed of constantly moving subatomic particles, McGarey asserts that, "All substances, then, whether they be living or not, give off vibrations . Is it really vibration, then, that carries the healing nature of castor oil into the body .?" (McGarey, 1993, p. 30). If this is the case, the question naturally arises as to how one measures the vibratory effects of a castor oil pack. References Callan JP. Holistic health or holistic hoax? [editorial]. JAMA.
1979; 241:1156.
Appendix A: Instructions For Castor Oil Packs To Make The Pack Take the piece of wool flannel and fold it into 3 thicknesses. Put it in a pan (like a large disposable baking pan) and pour castor oil on it. Saturate the whole flannel, and leave it until it is well-saturated. When you use it, you want it saturated, but not dripping. The size you want for use is about 12 x 18 inches - about the same size as your heating pad, so that the pad heats the whole pack, but does not touch your skin. After each use, you will probably need to add a little more castor oil. Where To Place The Pack Place the pack on the right side of the body, extending from a little above the bottom of the sternum (breastbone) to about 4 inches below the navel. It should go from the navel around the body on the right side as far to the side as you can get it. Basically, cover as much of your right side as you can. Using The Pack Use the pack in the evening, as you are resting before bed. Spread out a large plastic garbage bag on the bed so that the castor oil won't leak onto the bed. Fold a towel (that you will use only for castor oil packs, because the oil is almost impossible to wash out completely). Take the cloth cover off the heating pad. Then put the heating pad on top of the towel. Heat the pack before you put it on yourself. One way is to heat it in the oven on a "low" temperature setting. Another is to heat it in a microwave oven (in a microwave-safe container) for about one minute. Another way is to put the pack on top of the heating pad and turn the pad on high. Let it warm up for a few minutes. Lie down on your back on the plastic bag. Place the pack on your abdomen, with the heating pad on top and the towel on top of that. Have the heating pad control within easy reach of your hand, because you may need to adjust it so the pack is not too hot or too cold. It should be very warm, but not so hot it burns you. Be careful! If you're not sure how hot is safe, start with a slightly warm session and work up to a warmer one. If it's not warm enough, it may help to rest your arms and hands on the towel to press the pack onto your body. Keep the pack on for 1 to 1 ½ hours. Have a paper towel handy to wipe the oil off yourself when you get up, and be careful not to get any on the bed. Make up a solution of baking soda in warm water (2 teaspoons to a quart), and use paper towels or a sponge with the baking soda solution to clean off the castor oil. You might also want to take a shower with soap after that. Use the pack for 3 days in a row.
Appendix B: Tables 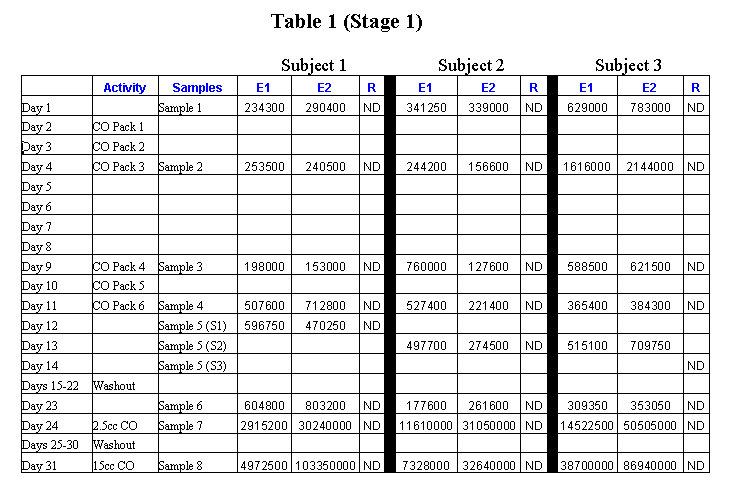 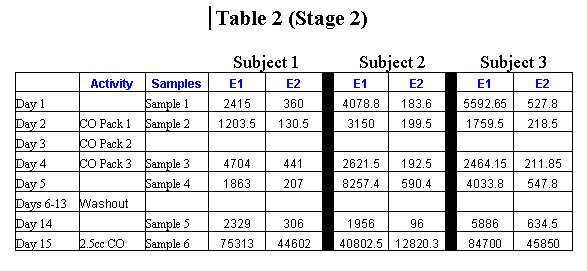 |