THERMOGRAPHIC ANOMALIES IN EPILEPSY PATIENTS
David McMillin, M.A., Douglas Richards, Ph.D., Eric Mein, M.D., and Carl Nelson, D.C.
Meridian Institute
[Click here to view this document in Adobe Acrobat (pdf) format.]
PURPOSE
The purpose of this study was to explore the feasibility of a hypothesis put forth by Edgar Cayce regarding the pathophysiology of epilepsy. Cayce stated that “From every condition that is of true [idiopathic] epileptic nature there will be found a cold spot or area between the lacteal duct and the caecum” (Cayce, 1934). Cayce’s explanation for the significance of such abdominal physiology is that “[vibration] is distributed to the body from that center of the body in solar plexus brain, or from those centers about the umbilicus, which are the electronic and atomic vibratory radiations of a human body” (Cayce, 1926).
Two specific objectives of this study were to collect preliminary data regarding Cayce’s abdominal epilepsy hypothesis that (1) could be gathered with a minimal budget; and (2) would be plausible to potential participating neurologists and epilepsy patients who might be recruited for a larger, controlled study should the initial evidence warrant further investigation.
TYPE OF STUDY
The project was an exploratory, descriptive study comparing abdominal thermograms of epilepsy patients with thermograms of patients with other conditions and healthy normals.
It utilized retrospective analysis of data. It involved no intervention.
SAMPLE
During the past 8 years we have evaluated 79 adults using liquid crystal thermography (LCT) for assessing abdominal thermal patterns. This data set includes diverse medical diagnoses (such as bowel disease, migraine, psoriasis, multiple sclerosis, chronic fatigue syndrome, and asthma) and healthy individuals. Included in this data are 6 epilepsy patients and a migraine patient with an epileptic-like EEG. All seven epilepsy patients were female adults.
PROCEDURE
Thermograms were made on Polaroid film using a Flexi-Therm LCT system. The subjects were lying on their backs on a chiropractic or massage table. Thermograms were made by placing the Flexi-Therm liquid crystal sheet on the exposed skin of the abdomen, and photographing the resulting pattern when it stabilized.
OUTCOME VARIABLES
The outcome variables in this study are variations in abdominal thermographic images in epilepsy patients as compared to controls (healthy normals and patients with other conditions).
DATA ANALYSIS
The abdominal LCT thermograms were via qualitative assessment by a team of researchers experienced in LCT. The goal was to determine whether there were visually apparent variations in the epilepsy patients with regard to increased coldness on the right side of the abdomen as compared to the left side in the epilepsy patients when compared to controls. Also, the assessment focused on possible thermal configurations that were relatively unique to epilepsy patients.
RESULTS
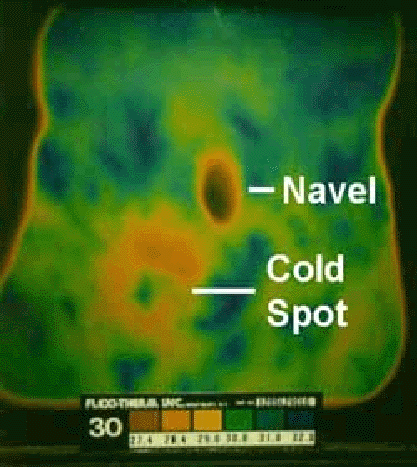
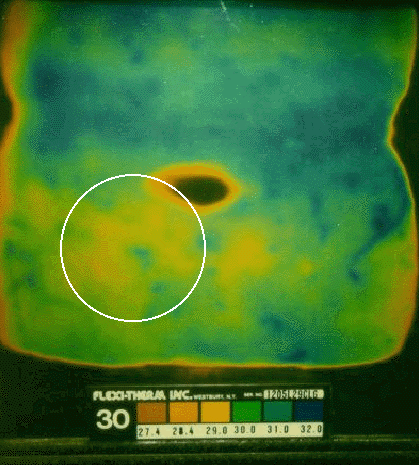
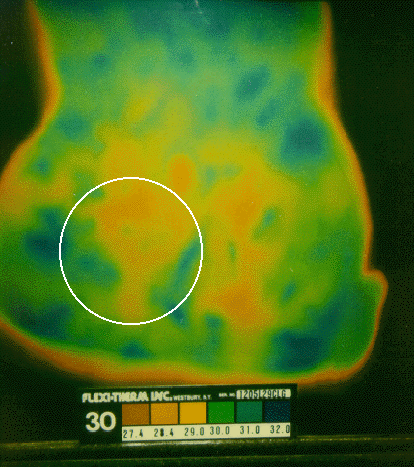
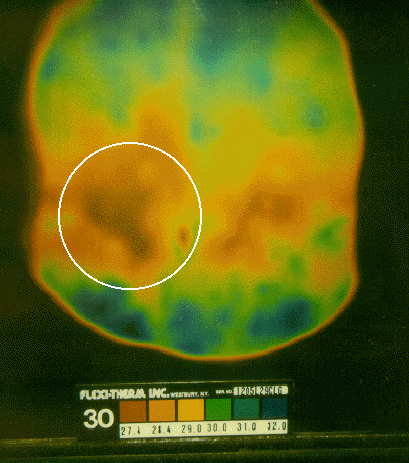
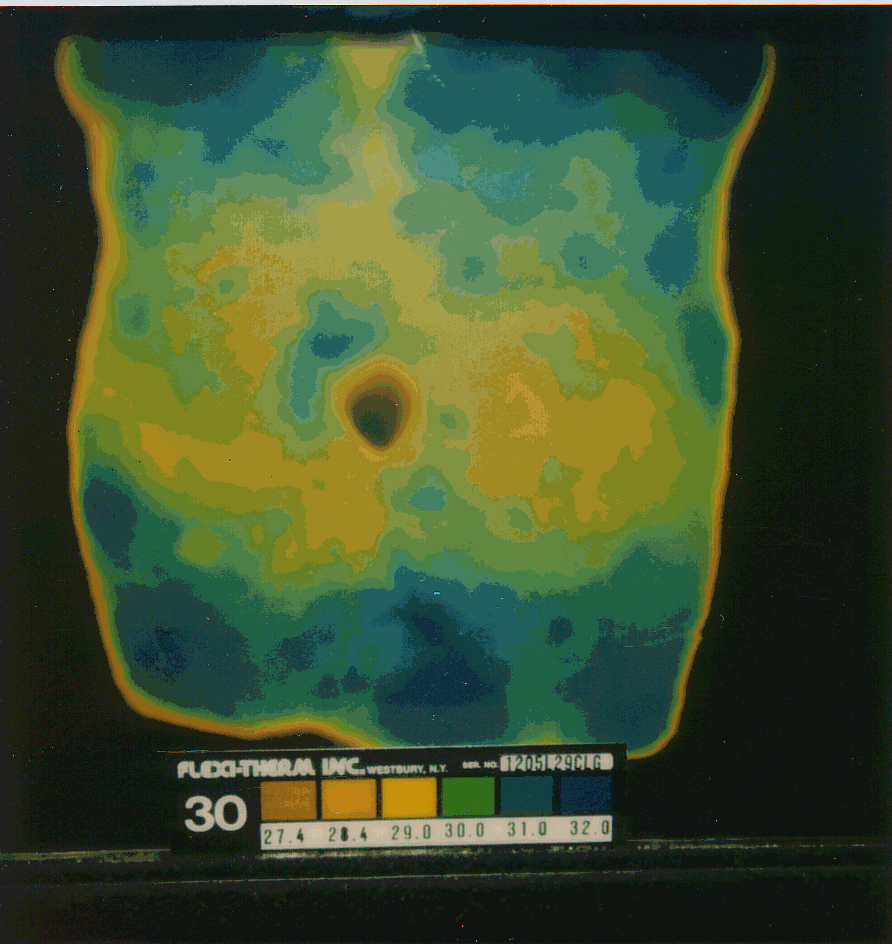
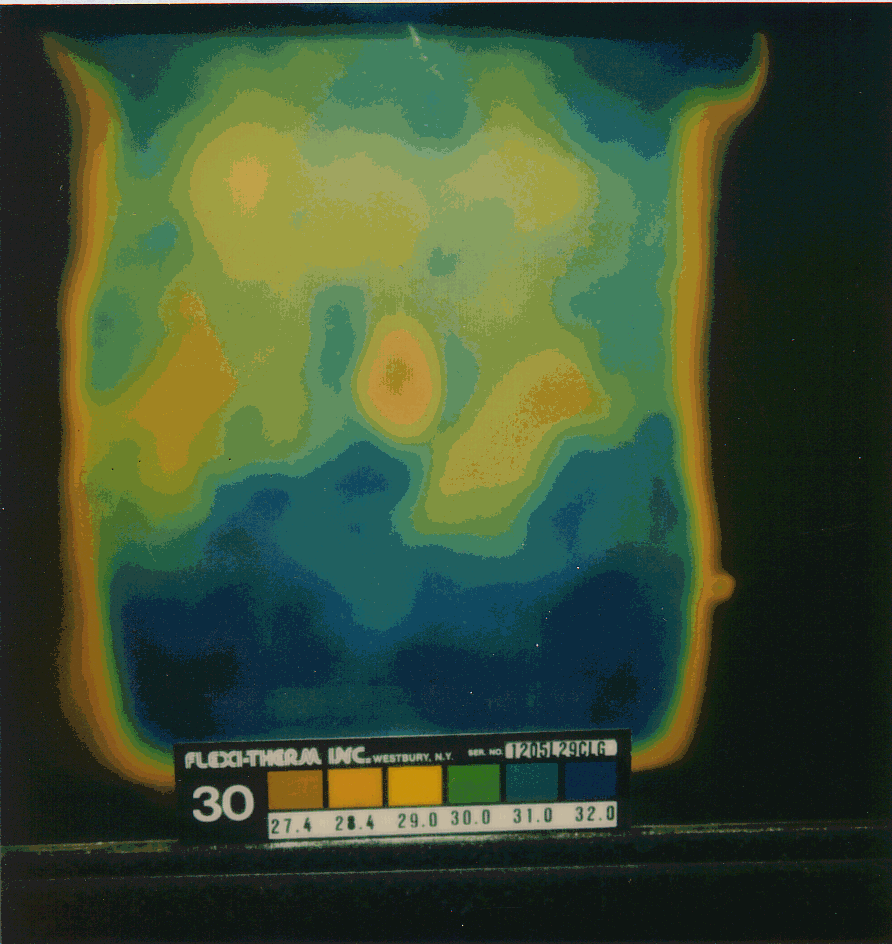
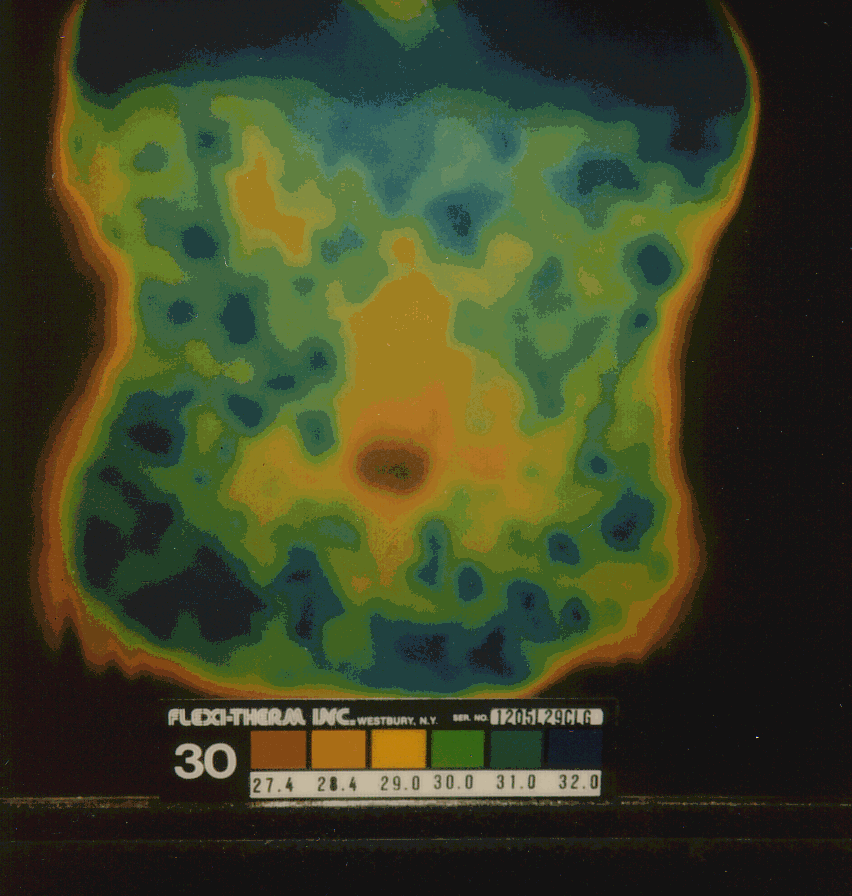
Abdominal thermograms of six epilepsy patients obtained using liquid crystal thermography (LCT) indicate a notable cold area on the right side of the abdomen as compared to the left side for each person. This pattern seems to be more common in epilepsy patients than with other illnesses or for healthy individuals. In four of the six cases of epilepsy the abdominal cold spot is a distinct pattern that occurs slightly below the navel. In two other cases the cool spot is less prominent and slightly above the navel on the right side. In contrast, in non-epileptic control subjects, no consistent pattern was noted. Table 1 provides graphic documentation of the four epilepsy cases with distinctive thermographic anomalies as compared to four nonepilepsy cases (2 normal, 1 asthma, 1 chronic fatigue syndrome).
DISCUSSION
Edgar Cayce, a prominent figure in the development of the holistic medicine movement in America, insisted that the etiology of most cases of idiopathic epilepsy could be traced to the peripheral nervous system, usually to the nerves of the abdomen. He also noted that abdominal thermal anomalies (especially a cold area on the right side of the abdomen) could be found in such cases (Cayce, 1934). This preliminary study supports the Cayce hypothesis.
The association of abdominal symptoms with epilepsy has been recognized for many years. For example, “gastric and intestinal disturbances” were viewed as primary etiological factors by medical doctors during the late 19th and early twentieth century (Musser and Kelly, 1912). Another example of the abdominal connection in epilepsy is the aura that is common in certain types of epilepsy. For example, temporal lobe epileptic seizures frequently begin with an aura. In neurological terms, an aura is actually a mild seizure that precedes the primary seizure. It can be thought of as a warning that a seizure is about to happen. Most often, auras manifest as an altered consciousness or peculiar sensation. “The most common aura is of vague gastric distress, ascending up into the chest” (Gordon, 1942, p. 610).
Modern medical science has rediscovered the abdominal connection in epilepsy. A few papers published in the medical journals during the 1960s called attention to the abdominal features in epilepsy. Our extensive review of the subject lists over 20 citations on the subject over the past twenty-five years (McMillin et al., 1999). Common clinical features of abdominal epilepsy include abdominal pain, nausea, bloating, and diarrhea with nervous system manifestations such as headache, confusion, and syncope (Peppercorn and Herzog, 1989). Although abdominal epilepsy is diagnosed most often in children, the research of Peppercorn and Herzog suggests that abdominal epilepsy may be more common in adults than generally realized.
One of the primary problems in understanding abdominal epilepsy is clearly defining the relationship of the abdominal symptoms to the seizure activity in the brain. Is the essential pathology in certain areas of the brain that happen to be connected to the abdominal organs? Or, is the primary pathology in the abdomen, which is conveyed through connecting nerve fibers to the brain, resulting in epileptic seizures? Peppercorn and Herzog noted both possibilities (1989), citing the vagus nerve as a logical source of nerve impulse between the brain and the gut.
The vagal link in epilepsy has also received attention with regard to a surgical procedure in which a pacemaker is implanted on the vagus nerve in the upper chest. Regular stimulation of the vagus has reduced or eliminated seizure activity in some treatment-resistent patients (e.g., Lundgren et al., 1998). If stimulation of the peripheral nervous system, in this case the vagus, can reduce seizure activity in the brain, perhaps pathological irritation of this or other peripheral nerves may also play a role in the etiology of certain forms of epilepsy.
Reflex epilepsy provides the key to understanding possible abdominal nerve involvement in the etiology of epilepsy. Reflex epilepsy includes a group of epileptic syndromes in which seizures are induced by a stimulus that may be simple (e.g., visual, somatosensory, olfactory, auditory) or complex (e.g., eating, thinking, reading). Most cases of documented reflex epilepsy are diagnosed because the triggering stimulus is easily identified. Visceral stimuli that leave a vasomotor signature could serve as reflex stimuli that are not as obvious as the typical reflex precipitants of seizure. We may be detecting such visceral reflex patterns in our thermograms.
One of the important questions that we have about our tentative findings is how to explain the underlying physiology that can link abdominal thermal variations to the etiology or pathophysiology of epilepsy. The ANS vasomotor system represents the leading candidate for connecting cutaneous thermography to neurologic illness. Thermography of biological systems assesses the status of dermal thermoregulatory function. The hypothalamus is believed to be the primary regulator of core temperature for body-heat maintenance (Bentzinger, 1969). This is essentially a systemic process by which the organism coordinates body temperature with internal (e.g., fever) and external (e.g., room temperature) factors and conditions. More precise local thermoregulation within each spinal dermatome may account for changes in dermal temperatures along the torso and extremities (Simon, 1975). This is generally described as a vasomotor process by which the sympathetic (SNS) division of the ANS regulates blood flow by dilating or constricting arterioles and capillaries within the innervated dermatome. Vasodilation tends to increase dermal temperature while vasoconstriction has the opposite effect. Thus the hypothalamic systemic regulation of temperature serves as a background against which location variations in ANS vasomotor function are manifested. We may have detected such local ANS vasomotor variations in our epilepsy patients.
Normal body surface temperatures are essentially symmetrical. Bilateral cutaneous thermal asymmetries and anomalies are suggestive of ANS vasomotor dysfunction, unless an identifiable surface feature (such as scar tissue or adhesion) is present to account for the variation. Likewise thermal anomalies in adjacent dermatomes are regarded as suggestive of sympathetic neuronal dysfunction (Korr, 1962). Some theorists maintain that SNS vasomotor function is closely associated with SNS visceromotor functioning. Thus thermographic anomalies may represent a “window on the sympathetic system” (Abernathy, 1988). With regard to the thermographic anomalies in our epilepsy data, the cold spot on the right side of the abdomen may represent an underlying ANS dysfunction that appears to be distinctive for that disorder within our limited sample. The ANS dysfunction could help account for the common abdominal/visceral symptoms within one or more subgroups of epilepsy.
In summary, the thermographic anomalies that we have measured in a small group of epilepsy patients may be linked to epileptic phenomena relating to the viscera (e.g., reflex epilepsy, visceral premonitions and auras, abdominal epilepsy, and vagus nerve involvement). If these findings are valid, the data could advance our understanding of the etiology of a physiologically distinct subgroup of epilepsy in which innovative treatment options directed at the peripheral nervous system and visceral organ systems may be developed as complementary and alternative medicine (CAM) options. Future studies will be required to validate these tentative findings. Based on the preliminary data, investment in more expensive equipment (digital infrared camera and software) and recruitment of a much larger sample are warranted.
REFERENCES
Abernathy M. Thermography: a window on the sympathetic nervous system. Thermology 1988; 1:4,5.
Bentzinger, TH, Heat regulation: Homeostasis of central temperature in man. Physiological Reviews, The American Physiological Society 1969; 49(4)671-752.
Cayce E. Edgar Cayce reading 1800-15; 1926.
Cayce E. Edgar Cayce reading 567-4; 1934.
Costa M, Brookes SJ. The enteric nervous system. Am J Gastroenterol. 1994;89:S29-137.
Gordon, B. (Ed.). Hughes’ Practice of Medicine. 16th ed. Philadelphia: The Blakiston Company; 1942:555-559.
Korr, IM, et. al. Effects of experimental myofascial insults on cutaneous patterns of sympathetic activity in man. Journal of Neural Transmission 1962; 23:22:330-355.
Lundgren J, Amark P, Blennow G, Stromblad LG, Wallstedt L. Vagus nerve stimulation in 16 children with refractory epilepsy. Epilepsia. 1998;39:809-813.
McMillin DL, Richards DG, Mein EA, Nelson CD. The abdominal brain and enteric nervous system. The Journal of Alternative and Complementary Medicine. 1999; 5(6):575-586.
Musser JH, Kelly AO. A Handbook of Practical Treatment. Philadelphia, PA: W. B. Saunders Co. 1912.
Peppercorn MA, Herzog AG, Dichter, MA & Mayman CI. Abdominal epilepsy: A cause of abdominal pain in adults. JAMA. 1978;40:2450-2451.
Peppercorn MA, Herzog AG. The spectrum of abdominal epilepsy in adults. Am J Gastroenterol. 1989;84:1294-1296.
Simon EK. Temperature regulation – spinal cord as a site of extrahypothalamic thermoregulation functions. Reviews of Physiology, Biochemistry and Pharmacology 1975;71:1-76.
 |
 |
 |
 |
 |
 |
 |
 |
